Section E
APPLICATION OF STEM PRINCIPLES AND PRACTICES
Due to the nature of the project, the application of Physics, Chemistry, and Math played a key role in the development of the final product. For the carrier, measurements of our SRO have been essential and the materials chosen have been essential for the production of the solution. The STF required an understanding of chemistry and the process of making the STF, and various volume, surface area, and other measurements were needed to apply the STF to the panels. The ballistic testing required an understanding of ballistic physics, especially with kinetic energy and the force a bullet exerts on surfaces.
V.1 Officer Culp Dimensions
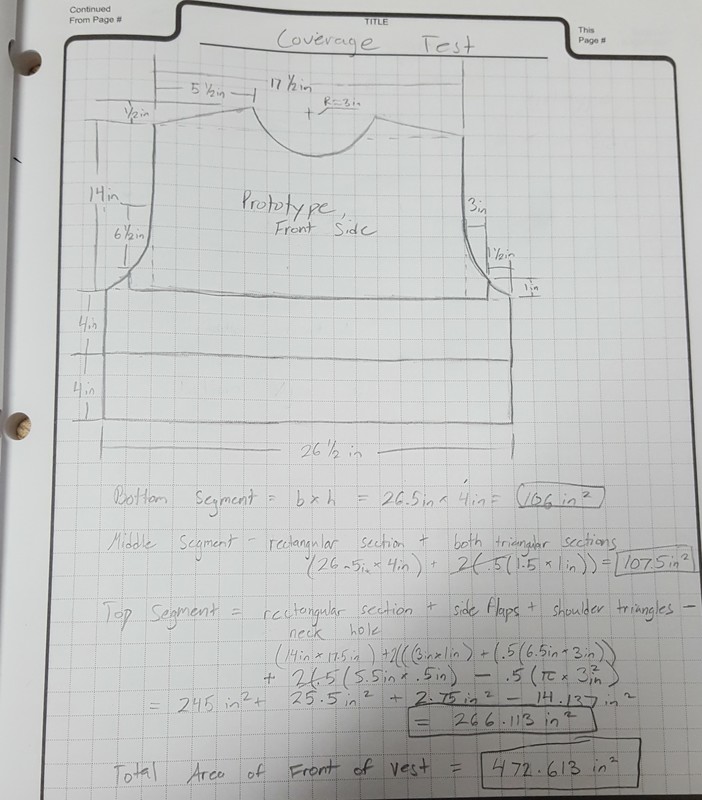
In order to make a vest that properly fits a user, measurements are made of various areas of the user. This is standard practice for body armor carrier companies, they send people to police departments whenever officers get new vests. In order to ensure that the design would fit Officer Culp and therefore would be comfortable, the team had to measure him. These measurements are shown below.
V.1 Officer Culp Dimensions
In order to make a vest that properly fits a user, measurements are made of various areas of the user. This is standard practice for body armor carrier companies, they send people to police departments whenever officers get new vests. In order to ensure that the design would fit Officer Culp and therefore would be comfortable, the team had to measure him. These measurements are shown below.
V.2 STF Research
The team knew the a significant portion of the project was to be dedicated to the shear thickening fluid. The team had to determine what particles were going to be suspended and what fluid they would be suspended in. Prior liquid body armor attempts used silica nanoparticles and then suspended them in polyethylene glycol. At that time, it was assumed that silica nanoparticles could not be bought online, so the team believed that they would have to be produced by the group. This lead the team to research how to produce the particles. The team sifted through numerous research papers, evaluating them based on time to manufacture, equipment needed, difficulty to manufacture, cost, and various other factors. The conclusion was that this paper contained the best manufacturing process for the team’s needs. It contained a relatively simple process, did not require equipment that could not be obtained, was relatively cheap, and was environmentally friendly. Although this process turned out to be a legitimate method of producing silica nanoparticles, the team had to make many assumptions when the first batch was made, as the process was quite vague. The team also was not sure how well the process would scale up, but it scaled up fine. The team concluded that this process could be industrialized, as rice hulls can be purchased in bulk for 6 dollars a ton (they are a waste product), most of the materials are available in large quantities for cheap, and most of the equipment used have large-scale industrial versions of them.
After the team determined how to make the silica nanoparticles, the next step was to figure out how to make a STF using the nanoparticles. It was known that the particles would be suspended in polyethylene glycol (PEG), but a ratio of silica to PEG was necessary. Some papers, like this paper, contained a weight percentage. The ideal ratio was a weight of silica to volume of PEG ratio, as liquids are easier to measure in volume. This ratio was found in this paper, so the 22 grams of silica to 78 mL of PEG ratio was used to create the STF.
Around a month after the team started producing the silica nanoparticles, the team researched some more about STF production and determined there were much easier ways to produce STFs. Instead of making colloidal silica like the group was manufacturing, these processes took fumed silica that one could purchase online and mixed that with the PEG to make a STF. This video shows this homemade STF, and this video even contains a link to how to make the STF. The team was disappointed that this process wasn't found earlier, but the group couldn’t back out of the rice hull process after the time and money that was put into it. However, it was decided that it was beneficial to purchase some fumed silica, mix it with PEG and see if it made a STF. Part of the reason the team decided to do this was because of this newly discovered this thesis. The entire thesis is dedicated to the creation of STFs and their uses in body armor. This paper provided an abundance of data that could be used to verify that the correct materials and proportions of them were used. The team ran into a problem when purchasing fumed silica in response to the thesis, the group didn’t know exactly what to purchase. The only hint was from the thesis, which said the fumed silica was purchased from Cabot Corporation USA. Their fumed silica is marketed as Cabosil, so the search was narrowed with that information. Looking on their website, there were many different types of fumed silica. The team didn’t know for sure which one to purchase, and even if that was the case, most Cabosil fumed silica available online wasn’t labeled with what type it was. couldn’t purchase it directly from the company in such small quantities, and this would have required the group to know what to purchase. With the team’s limited knowledge, this was purchased, which turned out to not produce a STF. Further research such as emailing the author of the thesis would help to determine which fumed silica to purchase.
V.3 STF Manufacturing
This section contains the outline of the silica nanoparticle production process, which is copied from the paper. Comments from the team are included to expand or replace information that pertains to this specific scenario. This process is accompanied with pictures from the team’s time at MTSU.
The collected rice husks were first washed with tap water to remove dirt and impurities and then dried in the oven at 60 ̊C for 24 h. The paper never mentions how to filter the materials, so steel window mesh was used in order to filter the impurities from the rice.)
(This part of the paper provides two methods of silica extraction. The rice hulls were pretreated with sulfuric acid because it provided the group with an ash comprising of a high percentage of silica and sulfuric acid was easily available. Rice hulls were treated with 1N (.5M) of sulfuric acid. The paper doesn’t say to wash the rice husks before combusting them, but that occurred in order to avoid the creation of sulfur trioxide, which is toxic. In order to wash them, the mesh was used to prevent the rice hulls from falling out of the containers when draining.)
The combustion process was conducted in an electric furnace at 600 ̊C for 6 h.
Silica extracted from rice husk (5 g) was dispersed in 500 mL of 0.5M NaOH aqueous solution and heated at 100 °C for 4 h under vigorous stirring to dissolve silica and produce sodium silicate
The solution obtained was filtered to remove the non-reactive impurities (Once again, the paper didn’t say how or what the impurities were. The metal mesh couldn't be used as the particles were too small. Instead, a vacuum filtration solution was used). The transparent filtrate of sodium silicate solution was allowed to cool to room temperature and titrated with 10% H2SO4 to pH 7 under vigorous stirring. Sodium silicate has shown to be neutralized with diluted sulfuric acid to precipitate silica (Lu and Hsieh 2012). Hereafter, the solution was first stirred for 24 h and then aged for 48 h to allow the silica gel to slowly precipitate.
The formed gel was fragmented (by poking it with a stir rod),
filtered (with the vacuum filtration), and washed with water to remove the sulfate salt. The clean silica gel was freeze-dried overnight to remove water.
The chemical reaction for this process, which is derived from above:
Na2SiO3 + H2SO4 ⇒ SiO2 + Na2SO4 +H2O
V.4 Kevlar Testing
In order to verify that the fluid was a shear thickening fluid, the team had to test its properties. The team decided to perform a stab test because it would utilize Kevlar and the knife would provide a high shear rate to the treated Kevlar. There were two panels, a treated panel with 4 layers of Kevlar and an untreated panel with 4 layers of Kevlar. The test simulated the testing conditions of NIJ 115.00 as accurately as the group could make it. This means dropping the knife so it impacts with a certain kinetic energy, which is 50J.
The team needed to figure out from what height to drop the knife from in order to achieve an impact with 50J of energy. To accomplish this, the formula for gravitational potential energy was used (air resistance was negligible). The drop tube apparatus that the knife was part of had a mass of 2.355kg and gravity was 9.8 m/s^2.
GPE = mgh
50J = 2.355kg * 9.8 m/s^2 * h
Height = 2.17m or 7.11ft
The formula for kinetic energy can be used to determine the velocity the knife will impact the Kevlar with.
KE = 1/2mv^2
50J = ½ * 2.355kg * v^2
Velocity = 6.52 m/s or 14.56 mph
After the STF was applied to the panel, the team could perform the main ballistic test.
The ballistic panels that the team tested were rated for Type IIA ballistic protection. The requirements for Type IIA protection are taken from NIJ Standard 101.06.
The kinetic energy formula is ½ mv^2 with m = mass in kilograms and v = velocity in meters per second. Although the group faced a drawback in which the vest was not new but was never worn, it can still be assumed that the panels would have to withstand a kinetic impact of between 504.1 J and 724.8 J.
.40 S&W FMJ 11.7g 352 m/s = ½ * (11.7g * (1kg/1000g)) * (352 m/s)^2 = 724.8J
9mm FRJ RN 8.0g 355 m/s = ½ * (8.0g * (1kg/1000g)) * (355m/s)^2 = 504.1J
The reason that fired bullets are so deadly is that they exert a significant force over a small area. Since force = mass * acceleration and the bullets are deaccelerated incredibly quickly, the Kevlar has to exert a strong force on the bullet to slow it down.To compound on this, the bullet exerts a significant pressure. Since pressure = force / area and the bullet has an incredibly small surface area, the bullet will exert a high amount of pressure. Bullets often have noses with small surface areas and are often pointed, not only because of aerodynamic efficiency but also to exert the highest possible pressure onto the surface which the gun is fired at. To illustrate this, a 20lb weight dropped from 8.15m will have the same kinetic as the .40 S&W FMJ 11.7g 352 m/s (and a momentum that is almost 30 times as large), the force of the weight is dispersed over a larger area. Since the force is exerted over a larger area, the impact of the weight dropping onto someone would hurt, but the weight wouldn’t puncture into his or her body (the bullet obviously does). Kevlar has a high tensile strength that can withstand the pressure exerted by the bullet.
To figure out the pressure that Type IIA armor has to withstand, the team can use data from the NIJ standards to figure out the pressure. Work = force * distance, so 724.8J / .00635m (the depth from our tests) = 114141N of force. The area of the nose of the .40 S&W is approximately equal to the radius of the top of the nose squared and that multiplied by pi. The nose diameter is 10.2mm. So 114141N / ((10.2mm * (1m/1000mm))^2 * pi) equals 350MPa. With this same calculation (with some approximations involving kinetic energy) , our .22 Long Rifle round exerted a pressure of ((191J/.00625m)/((.0057m)^2 * pi) = 295MPa. For reference, the tensile strength of skin according to this paper is around 25MPa
.40 S&W FMJ 11.7g 352 m/s = ½ * (11.7g * (1kg/1000g)) * (352 m/s)^2 = 724.8J
9mm FRJ RN 8.0g 355 m/s = ½ * (8.0g * (1kg/1000g)) * (355m/s)^2 = 504.1J
The reason that fired bullets are so deadly is that they exert a significant force over a small area. Since force = mass * acceleration and the bullets are deaccelerated incredibly quickly, the Kevlar has to exert a strong force on the bullet to slow it down.To compound on this, the bullet exerts a significant pressure. Since pressure = force / area and the bullet has an incredibly small surface area, the bullet will exert a high amount of pressure. Bullets often have noses with small surface areas and are often pointed, not only because of aerodynamic efficiency but also to exert the highest possible pressure onto the surface which the gun is fired at. To illustrate this, a 20lb weight dropped from 8.15m will have the same kinetic as the .40 S&W FMJ 11.7g 352 m/s (and a momentum that is almost 30 times as large), the force of the weight is dispersed over a larger area. Since the force is exerted over a larger area, the impact of the weight dropping onto someone would hurt, but the weight wouldn’t puncture into his or her body (the bullet obviously does). Kevlar has a high tensile strength that can withstand the pressure exerted by the bullet.
To figure out the pressure that Type IIA armor has to withstand, the team can use data from the NIJ standards to figure out the pressure. Work = force * distance, so 724.8J / .00635m (the depth from our tests) = 114141N of force. The area of the nose of the .40 S&W is approximately equal to the radius of the top of the nose squared and that multiplied by pi. The nose diameter is 10.2mm. So 114141N / ((10.2mm * (1m/1000mm))^2 * pi) equals 350MPa. With this same calculation (with some approximations involving kinetic energy) , our .22 Long Rifle round exerted a pressure of ((191J/.00625m)/((.0057m)^2 * pi) = 295MPa. For reference, the tensile strength of skin according to this paper is around 25MPa